Industry Insights: Bioconjugate innovation gains momentum across partnerships and pivotal trials
Bioconjugate Insights 2026; 1(1), 49–66 ·
DOI: 10.18609/bci.2026.008
| February saw a sustained pace of partnering and portfolio reshaping across the bioconjugate space, alongside multiple late-stage clinical and regulatory catalysts. On the dealmaking front, Chugai exercised its option to license Araris’ multi-payload linker-payload platform, while Wave regained full rights to its GalNAc-conjugated RNA-editing therapy from GSK and signaled plans to accelerate US FDA engagement. Collaboration activity also broadened beyond classic ADCs with HealZen initiating a cross-border partnership in degrader-antibody conjugates, StarkAge teaming with Gustave Roussy to support senolytics ADC development, and Rakuten Medical signing LOTTE Biologics for the manufacturing of antibody-dye photoimmunotherapy conjugates. Multiple platform and asset licenses underscored continued demand for differentiated formats, including B7-H3 ADC and bispecific/dual-payload ADC agreements with MediLink-Roche and Biocytogen-Acepodia, and proximity-guided multispecific work with InduPro-Lilly. Regulators advanced several programs, including Japan supplement NDA submissions for DATROWAY and PADVEC plus pembrolizumab, and China approval of ENHERTU in second-line HER2-positive gastric/gastroesophageal junction adenocarcinoma cancer. Clinically, first-patient dosing milestones spanned HER2, CD37, PD-L1, CDH17, and PSMA-targeted conjugates, while OPUS-3 initiated evaluation of VAX-31 in previously vaccinated adults. Market activity included Sutro’s $110M financing and Intensity’s reverse split, reflecting continued capital management amid ongoing development. |  Rakuten Medical and LOTTE Biologics signed manufacturing agreement for photoimmunotherapy bioconjugates; Collaborations, Partnerships, and Acquisitions. Credit: www.lottebiologics.com |
COLLABORATIONS, PARTNERSHIPS, AND ACQUISITIONS |
Chugai exercised option to license Araris Biotech AG’s multi-payload ADC platform [1]
Chugai Pharmaceutical announced that it exercised its option under a previously established Research Collaboration and Option to License agreement with Araris Biotech to access the proprietary AraLinQ® linker–payload platform for development of ADCs against one selected target. AraLinQ® is designed to enable conjugation of multiple anticancer payloads to a single antibody with antibody-like pharmacokinetics and linker stability. Under the agreement, Araris will receive an upfront payment and is eligible for milestone payments and royalties on potential commercial sales. The broader collaboration provides for total potential consideration of up to $780M, contingent on option exercises and milestone achievements.
SK bioscience launched MenACWY-TT conjugate vaccine in Korea through Sanofi partnership [2]
SK bioscience announced the domestic launch of MenQuadfi (MenACWY-TT), a quadrivalent meningococcal conjugate vaccine developed by Sanofi, for the prevention of invasive meningococcal disease caused by serogroups A, C, W, and Y. The tetanus toxoid–conjugated vaccine is approved in Korea for individuals aged 6 weeks to 55 years. It is the only MenACWY vaccine in the country indicated for infants aged 6 weeks to <24 months that includes serogroup A coverage. SK bioscience will oversee domestic distribution for infant and pediatric populations. MenQuadfi is supplied as a fully liquid 0.5mL intramuscular injection, eliminating the need for reconstitution and supporting streamlined clinical administration.
Wave regained full rights to GalNAc-conjugated RNA editing therapy WVE-006 from GSK [3]
Wave Life Sciences announced that it has regained full control of WVE-006, a GalNAc-conjugated oligonucleotide RNA editing therapy for alpha-1 antitrypsin deficiency (AATD), from partner GSK. WVE-006 is designed to correct the Z mutation in SERPINA1 mRNA, restoring functional M-AAT protein while reducing toxic Z-AAT accumulation. In the Phase 1b/2a RestorAATion-2 trial, repeat 200mg doses increased M-AAT to 7.2 µM and reduced circulating Z-AAT by approximately 60%, while a single 400mg dose increased total AAT to 12.8 µM. Higher-dose cohort data are expected this year. Wave plans to engage the FDA regarding a potential accelerated approval pathway, with regulatory feedback anticipated by mid-2026.
StarkAge Therapeutics entered research collaboration with Gustave Roussy to support development of senolytic ADC [4]
StarkAge Therapeutics announced a research collaboration with Gustave Roussy to characterize cellular senescence induced by standard-of-care treatments across digestive cancers. The findings are intended to inform clinical development of STX-01, a first-in-class senolytic ADC targeting DDP4, a marker of senescence that is frequently expressed on primary tumors and/or overexpressed in metastases. The program will combine StarkAge’s expertise in senescence biology with Gustave Roussy’s capabilities in multiplexed pathology and computational oncology under a research agreement with Gustave Roussy and Gustave Roussy Transfert. The collaboration is expected to support IND-enabling studies and refine the positioning of senescence-targeted ADC strategies in oncology.
HealZen entered strategic partnership to advance degrader–antibody conjugate therapeutics [5]
HealZen Therapeutics announced a strategic partnership with a US biotechnology company to advance degrader–antibody conjugate (DAC) therapeutics, described as its first cross-border collaboration for this modality. Under the agreement, HealZen will retain Greater China rights to the partnered DAC programs and is eligible to receive an upfront payment, milestone payments, tiered royalties on sales, and potential sublicense income. The collaboration expands HealZen’s activities in targeted protein degradation and follows a previously announced out-licensing agreement for a Bruton’s tyrosine kinase degrader with Johnson & Johnson. Further details of the DAC programs and development plans were not disclosed and will be announced at a later date.
Rakuten Medical and LOTTE Biologics signed manufacturing agreement for photoimmunotherapy bioconjugates [6]
Rakuten Medical and LOTTE Biologics entered a biopharmaceutical manufacturing agreement to support production of Rakuten Medical’s Alluminox® platform-based photoimmunotherapy. LOTTE Biologics will provide manufacturing services for monoclonal antibody intermediates and their conjugates at its ADC-capable Syracuse Bio Campus facility in New York. The agreement supports global clinical development and future commercialization, including an ongoing Phase 3 trial in head and neck cancer and planned expansion into additional solid tumors. Rakuten Medical’s therapy consists of a cell-targeting antibody conjugated to a light-activatable dye (IRDye® 700DX), enabling selective tumor cell destruction upon localized illumination. The collaboration strengthens commercial-scale supply of these antibody–dye bioconjugates.
Tahoe and Alloy formed joint venture to develop first-in-class ADCs against novel tumor targets [7]
Tahoe Therapeutics and Alloy Therapeutics formed a jointly seeded company to develop first-in-class ADCs targeting novel tumor-specific surface antigens identified through Tahoe’s Mosaic platform and large-scale single-cell datasets. The new venture will initially advance two ADC programs based on targets discovered using Tahoe’s AI-powered virtual cell models and validated across independent assays and clinical samples. Tahoe will contribute target discovery and biomarker insights, while Alloy will provide ADC engineering capabilities, translational expertise, and venture studio support through 82VS. The companies will co-invest and co-lead the entity, aiming to progress both programs to value-inflection points suitable for independent financing or pharmaceutical partnerships.
Summit Therapeutics and GSK entered clinical collaboration to evaluate PD-1/VEGF bispecific with B7-H3-targeted ADC [8]
Summit Therapeutics entered a clinical trial collaboration with GSK to evaluate ivonescimab, a PD-1/vascular endothelial growth factor (VEGF) bispecific antibody, in combination with risvutatug rezetecan (GSK’227), a B7-H3-targeted ADC, across multiple solid tumors, including small cell lung cancer (SCLC). The study will assess safety and preliminary antitumor activity, with patient dosing expected to begin in mid-2026. Summit will supply ivonescimab, while GSK will oversee clinical operations; both parties retain rights to their respective products under a non-exclusive agreement. Risvutatug rezetecan comprises a fully human anti-B7-H3 monoclonal antibody linked to a topoisomerase inhibitor payload and is currently in a Phase 3 trial in relapsed extensive-stage SCLC.
AbbVie entered exclusive licensing agreement with RemeGen for PD-1/VEGF bispecific antibody [9]
AbbVie and RemeGen entered an exclusive licensing agreement for RC148, an investigational PD-1/VEGF-targeted bispecific antibody being evaluated in advanced solid tumors. AbbVie gains rights outside Greater China, while RemeGen will receive $650M upfront and is eligible for up to $4.95B in development, regulatory, and commercial milestones plus tiered double-digit royalties. RC148 is being studied as monotherapy and in combination regimens, including ADC combinations, and has shown preliminary antitumor activity in early clinical studies. The collaboration enables potential combination strategies with AbbVie’s ADC portfolio, including investigational telisotuzumab adizutecan, across indications such as non-SCLC (NSCLC) and colorectal cancer.
Biocytogen and Acepodia entered option and license agreement for dual-payload bispecific ADC programs [10]
Biocytogen and Acepodia entered an option and license agreement to evaluate and advance dual-payload bispecific ADC (BsADC) programs. The agreement grants Acepodia an option to obtain an exclusive worldwide license to two BsADC candidates, with Biocytogen eligible for an upfront option fee, option exercise fees, development, regulatory, and commercial milestones, and royalties on future sales. The collaboration combines Biocytogen’s RenLite® platform for fully human bispecific antibody discovery with Acepodia’s Antibody-Dual-Drugs Conjugation (AD2C) technology to develop next-generation dual-payload BsADCs. The partnership builds on prior co-development activities and aims to evaluate candidate selection and advancement based on ongoing preclinical research and defined governance criteria.
MediLink entered exclusive global licensing agreement with Roche for B7H3-targeted ADC [11]
MediLink Therapeutics entered a collaboration and exclusive licensing agreement with Roche for the development and commercialization of YL201, a B7H3-targeted ADC. MediLink will receive $570M in upfront and near-term milestone payments, plus additional development, regulatory, and commercial milestones and tiered royalties on net sales outside of China. Roche gains worldwide rights excluding mainland China, Hong Kong, and Macau. YL201, developed using MediLink’s Tumor Microenvironment-Activatable LINker-payload platform, is in multinational clinical trials for advanced solid tumors and is in Phase 3 registrational studies in China for SCLC and nasopharyngeal carcinoma. The FDA previously granted Breakthrough Therapy Designation for YL201 in SCLC, along with three Orphan Drug Designations.
InduPro entered partnership with Eli Lilly to advance proximity-based BsADCs [12]
InduPro Therapeutics entered a collaboration with Eli Lilly valued at up to $950M to explore up to three oncology targets using InduPro’s proximity-guided platform. The approach identifies tumor-associated proximity antigens (TAPAs), surface proteins spatially co-localized with tumor-associated antigens, to enable selective co-targeting in bispecific and multispecific therapeutics, including ADCs and T cell engagers. Lilly gains access to InduPro’s AI and machine learning-enabled membrane interactomics platform, while InduPro will lead early discovery and co-target identification; Lilly will also make an equity investment. InduPro’s lead asset, IDP-001, is a proximity-based BsADC targeting EGFR and a novel tumor-associated antigen, with an IND filing planned for Q1 2026.
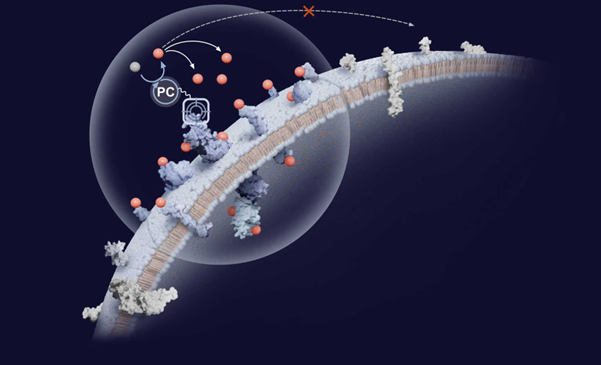 |
InduPro entered partnership with Eli Lilly to advance proximity-based BsADCs; Conferences, Partnerships, and Acquisitions. Credit: www.induprotx.com |
Amgen entered licensing agreement with DISCO to develop therapies against novel cancer surface target [13]
Amgen entered an exclusive global licensing agreement with DISCO Pharmaceuticals to develop and commercialize therapeutic programs directed against an undisclosed cancer cell surface antigen identified using DISCO’s surfaceome mapping platform. The deal could be worth up to $618M in milestone payments, in addition to sales royalties. DISCO’s platform combines cell-surface proteomics with protein community mapping to identify novel surface-associated target pairs, thereby supporting the development of modalities such as BsADCs and T cell engagers. While the specific therapeutic format was not disclosed, the agreement centers on translating newly identified surface targets into oncology candidates. DISCO recently raised $42M in seed financing to advance its lead ADC programs toward IND-enabling studies.
Synaffix and Sidewinder entered a multi-target license agreement to develop BsADCs [14]
Synaffix, a Lonza company, and Sidewinder Therapeutics announced a multi-target licensing agreement to develop BsADCs for solid tumor indications. Under the agreement, Sidewinder gains access to Lonza’s clinically validated, site-specific ADC platform, including GlycoConnect® antibody conjugation technology, HydraSpace® polar spacer technology, and a toxSYN® linker–payload. The collaboration aims to combine Sidewinder’s bispecific antibodies, designed for enhanced tumor-cell specificity and internalization, with Synaffix’s linker–payload technologies to generate first-in-class BsADCs with an improved therapeutic index. Lonza will receive upfront, clinical, regulatory, and commercial milestone payments, plus royalties on net sales. Lonza will manufacture components related to its proprietary technologies, while Sidewinder will lead research, development, manufacturing, and commercialization of the resulting ADCs.
OBI Pharma licensed glycan-targeting ADC to TegMine Therapeutics under global commercial agreement [15]
OBI Pharma entered a global commercial license agreement with TegMine Therapeutics for a glycan-targeting ADC developed using OBI’s Obrion™ platform technologies. Under the agreement, OBI will receive an upfront payment, development and commercial milestones, and tiered royalties on annual net sales. TegMine gains exclusive worldwide rights to develop and commercialize the ADC. The candidate combines a high-affinity anti-glycan antibody from TegMine with OBI’s site-specific GlycOBI® glycan conjugation, EndoSymeOBI® enzymatic technology, and HYPrOBI® hydrophilic linker to generate a homogeneous, scalable ADC. The collaboration advances glycan-targeted ADC development and reflects continued external partnering of OBI’s site-specific conjugation technologies.
REGULATORY CHANGES AND UPDATES |
Daiichi Sankyo submitted sNDA in Japan for datopotamab deruxtecan in first-line TNBC [16]
Daiichi Sankyo announced submission of a supplemental New Drug Application (sNDA) to Japan’s Ministry of Health, Labour and Welfare for datopotamab deruxtecan (DATROWAY®), a TROP2-directed DXd ADC, for adult patients with hormone receptor-negative, HER2-negative unresectable or recurrent breast cancer (triple-negative breast cancer [TNBC]). The application is based on the Phase 3 TROPION-Breast02 trial, which demonstrated statistically significant improvements in overall survival and progression-free survival versus investigator’s choice of chemotherapy in first-line metastatic TNBC patients ineligible for immunotherapy. TROPION-Breast02 enrolled 644 patients globally. DATROWAY comprises a humanized anti-TROP2 IgG1 antibody linked via tetrapeptide-based cleavable linkers to a topoisomerase I inhibitor payload (exatecan derivative, DXd).
Astellas submitted sNDA in Japan for enfortumab vedotin plus pembrolizumab in muscle-invasive bladder cancer [1]
Astellas announced submission of an sNDA to Japan’s Ministry of Health, Labour and Welfare for enfortumab vedotin (PADCEV), a Nectin-4-directed ADC, in combination with pembrolizumab for neoadjuvant treatment followed by adjuvant therapy in adults with muscle-invasive bladder cancer ineligible for cisplatin-based chemotherapy. The filing is based on the Phase 3 EV-303 (KEYNOTE-905) trial, which demonstrated a 60% reduction in risk of recurrence, progression, or death and a 50% reduction in risk of death versus standard management. The safety profile was consistent with prior reports, and no new safety signals were identified. The combination was approved in the US in November 2025, with European review ongoing.
Henlius received China NMPA IND approval for PD-L1-targeting ADC HLX43 in combination regimens [18]
Shanghai Henlius Biotech announced that China’s National Medical Products Administration approved the IND application for a clinical trial of HLX43, a PD-L1-targeting ADC, in combination with the anti-PD-1 monoclonal antibody serplulimab (HANSIZHUANG) and the anti-EGFR monoclonal antibody HLX07 for advanced solid tumors. HLX43 is designed to integrate immune checkpoint blockade with payload-mediated cytotoxicity. Early clinical data have indicated manageable safety and antitumor activity in multiple solid tumors, including NSCLC. The company is progressing dual-combination studies of HLX43 with serplulimab and with HLX07, with plans to expand into a triple-combination regimen to explore synergistic effects across solid tumor indications.
ENHERTU approved in China for second-line HER2-positive gastric or GEJ adenocarcinoma [19]
ENHERTU (trastuzumab deruxtecan [T-DXd]), a HER2-directed DXd ADC co-developed by Daiichi Sankyo and AstraZeneca, was approved by China’s National Medical Products Administration for adult patients with locally advanced or metastatic HER2-positive (IHC 3+ or IHC 2+/ISH+) gastric or gastroesophageal junction adenocarcinoma (GEJ) after one prior trastuzumab-based regimen. The approval was based on the Phase 3 DESTINY-Gastric04 trial, in which ENHERTU reduced the risk of death by 30% versus ramucirumab plus paclitaxel (hazard ratio 0.70; p=0.0044), with median overall survival of 14.7 versus 11.4 months. Progression-free survival and objective response rate were also improved. The safety profile was consistent with previous studies, with interstitial lung disease reported in 1.6% of patients as grade 5 events.
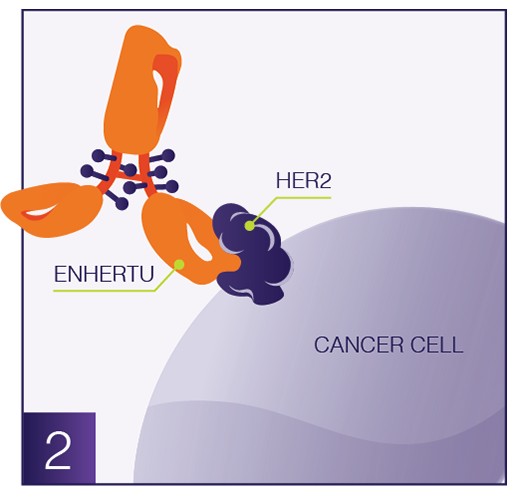 |
| ENHERTU approved in China for second-line HER2-positive gastric or GEJ adenocarcinoma; Regulatory Changes and Updates. Credit: www.enhertu.com |
NEOK Bio received FDA IND approval for bispecific ADC NEOK001 [20]
NEOK Bio announced that the FDA approved its IND application for NEOK001, enabling initiation of a Phase 1 clinical trial. NEOK001 is a bispecific ADC targeting B7-H3 and ROR1, two surface proteins reported to be highly expressed in cancer cells. The construct incorporates a topoisomerase I inhibitor payload linked to the bispecific antibody. Preclinical studies demonstrated superior in vivo efficacy in solid tumor models compared to monovalent ADCs. The upcoming Phase 1 trial will evaluate safety and clinical activity, with first patient dosing anticipated in the coming months and initial clinical data expected in 2027. NEOK001 is the first program from the company’s pipeline of bispecific ADCs to enter clinical development.
Avacta received FDA clearance of IND for FAP-Exd peptide-drug conjugate AVA6103 [21]
Avacta Therapeutics announced FDA clearance of IND application for AVA6103 (FAP-Exd), its second pre|CISION® tumor-activated oncology program and the first pre|CISION peptide drug conjugate incorporating the topoisomerase I (TOP 1) inhibitor exatecan. AVA6103 is designed to be activated in the TME through targeting fibroblast activation protein (FAP). The Phase 1 trial will assess safety, potential efficacy, and dose selection in adult patients with pancreatic, cervical, gastric, and SCLC. The dose-escalation study includes two parallel administration schedules of every 2 weeks and every 3 weeks. Tumor selection was informed by an artificial intelligence-based approach evaluating sensitivity markers for TOP1 inhibitors and FAP expression under a collaboration with Tempus AI. Preliminary data are anticipated in the second half of 2026.
EU approves donidalorsen, a GalNAc-conjugated ASO, for prophylactic treatment of hereditary angioedema [22]
Otsuka has received EU approval for Dawnzera® (donidalorsen) for routine prevention of recurrent hereditary angioedema (HAE) attacks in patients aged ≥12 years, following prior FDA authorization. Donidalorsen is a ligand-conjugated antisense oligonucleotide (ASO) designed to reduce hepatic prekallikrein production, thereby interrupting the kallikrein–bradykinin pathway underlying HAE attacks. The approval is based on Phase 3 OASIS-HAE data showing an 81% reduction in 4-week attack rate versus placebo with monthly dosing and a 55% reduction with dosing every 8 weeks. At week 24, 91% of patients receiving monthly therapy achieved well-controlled disease. Donidalorsen represents a clinically validated example of targeted oligonucleotide bioconjugation in rare disease therapeutics.
BPGbio received FDA orphan drug designation for BPM31510 in primary CoQ10 deficiency [23]
BPGbio announced that the FDA granted orphan drug designation to BPM31510 for the treatment of primary coenzyme Q10 (CoQ10) deficiency, an ultra-rare mitochondrial disorder affecting fewer than one in 100,000 individuals. BPM31510 is a CoQ10–lipid nanoparticle conjugate designed for intravenous administration to deliver supraphysiological levels of CoQ10 directly to mitochondria. The designation follows a previous Rare Pediatric Disease Designation. The company reported preliminary clinical data from multiple compassionate use cases presented at mitochondrial conferences and stated that development has proceeded with FDA engagement on an expedited pathway. The therapeutic rationale and candidate identification were supported by BPGbio’s NAi Interrogative Biology® platform, which applies Bayesian artificial intelligence to model biological data and mitochondrial pathway relationships.
FDA granted Breakthrough Therapy designation to Lilly’s FRα-targeted ADC sofetabart mipitecan in platinum-resistant ovarian cancer [24]
The FDA granted Breakthrough Therapy designation to sofetabart mipitecan (LY4170156) for adult patients with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer previously treated with bevacizumab and mirvetuximab soravtansine, if eligible. Sofetabart mipitecan is a folate receptor alpha (FRα)-targeted ADC incorporating proprietary linker technology and an exatecan payload. The designation is supported by preliminary Phase 1 data demonstrating responses across all FRα expression levels, including in patients previously treated with mirvetuximab, and a favorable tolerability profile. The program has advanced into the global Phase 3 FRAmework-01 trial evaluating the ADC as monotherapy and in combination with bevacizumab.
Dyne Therapeutics granted Orphan Drug designation to Dyne’s TfR1-targeted ASO–Fab conjugate z-basivarsen for DM1 [25]
Dyne Therapeutics announced that Japan’s Ministry of Health, Labour and Welfare granted Orphan Drug designation to zeleciment basivarsen (z-basivarsen) for myotonic dystrophy type 1 (DM1). Z-basivarsen is an ASO conjugated to a transferrin receptor 1 (TfR1)-binding Fab fragment to enable targeted delivery to muscle and the central nervous system. The therapy is being evaluated in the Phase 1/2 ACHIEVE trial, where early data have shown sustained improvements in myotonia, muscle strength, and function with a favorable safety profile. Z-basivarsen has also received Breakthrough Therapy, Fast Track and Orphan Drug designations in the US, and Orphan Drug designation in the EU.
EMA validated Type II variation for ENHERTU plus pertuzumab in first-line HER2-positive metastatic breast cancer [26]
The European Medicines Agency (EMA) validated a Type II variation application for ENHERTU in combination with pertuzumab for first-line treatment of unresectable or metastatic HER2-positive breast cancer. The submission is based on results from the Phase 3 DESTINY-Breast09 trial, in which ENHERTU plus pertuzumab demonstrated a statistically significant and clinically meaningful improvement in progression-free survival compared with taxane, trastuzumab, and pertuzumab (THP). ENHERTU is a HER2-directed DXd ADC jointly developed by Daiichi Sankyo and AstraZeneca. Validation initiates the scientific review by the EMA’s Committee for Medicinal Products for Human Use. ENHERTU recently received approval in the US for this first-line indication.
Whitehawk received FDA IND clearance for PTK7-targeted ADC HWK-007 and submitted IND for HWK-016 [27]
Whitehawk Therapeutics announced that the FDA cleared its IND application for HWK-007, a PTK7-targeted ADC, enabling initiation of a Phase 1 trial in advanced or metastatic solid tumors, including lung, ovarian, and endometrial cancers. The company also submitted an IND for HWK-016, a MUC16-targeted ADC, with Phase 1 initiation anticipated this quarter. Both programs utilize a next-generation ADC platform incorporating a stable yet cleavable linker, a DNA TOP1 inhibitor payload, and a carbon-bridge cysteine re-pairing bioconjugation strategy. The Phase 1 studies will employ dose-escalation and expansion designs to assess safety, pharmacokinetics, and preliminary antitumor activity, with initial clinical data expected in early 2027.
Arbele received FDA orphan drug designation for CDH17-targeting ADC ARB1002 [28]
Arbele announced that the FDA granted Orphan Drug Designation to ARB1002, an investigational ADC for pancreatic cancer. ARB1002 comprises an anti-CDH17 monoclonal antibody chemically linked to a cytotoxic payload and is designed to selectively target CDH17, a cell-surface adhesion molecule overexpressed in pancreatic tumors, to enable targeted delivery while limiting off-target exposure. The designation provides regulatory incentives for therapies addressing rare diseases affecting fewer than 200,000 patients in the US. Arbele has completed IND-enabling studies and plans to initiate a Phase 1 clinical trial this year to evaluate ARB1002 in patients with pancreatic cancer.
 |
| Arbele received US FDA orphan drug designation for CDH17-targeting ADC ARB100228; Regulatory Changes and Updates. Credit: depositphotos.com |
FibroGen announces rebranding to Kyntra Bio following new focus on novel therapies [29]
FibroGen announced its rebranding to Kyntra Bio and highlighted regulatory progress for its oncology and rare disease pipeline. Roxadustat received Orphan Drug Designation from the FDA for the treatment of myelodysplastic syndromes (MDS). In oncology, Kyntra Bio is advancing FG-3246, a CD46-targeting ADC, and FG-3180, a CD46-targeting positron emission tomography imaging agent. Topline results from a Phase 1b/2 study of FG-3246 in combination with enzalutamide in metastatic castration-resistant prostate cancer are expected in Q1 2026, with interim Phase 2 monotherapy data anticipated in H2 2026.
MARKET TRENDS |
Intensity Therapeutics approved 1-for-25 reverse stock split to regain Nasdaq compliance [30]
Intensity Therapeutics announced that its board approved a 1-for-25 reverse stock split of its common stock, effective February 18, 2026, to regain compliance with Nasdaq’s minimum bid price requirement. The reverse split will reduce outstanding shares from approximately 63,346,579 to approximately 2,533,863, while the total authorized share count will remain unchanged. Proportional adjustments will be made to outstanding stock options, warrants, convertible securities, and equity incentive plans. Intensity develops intratumoral cancer therapies using a proprietary non-covalent conjugation technology platform, and the company’s shares will continue trading on The Nasdaq Capital Market under the ticker ‘INTS’ on a split-adjusted basis beginning February 19, 2026.
Sutro priced $110M public offering to support ADC pipeline development Sutro [31]
Sutro Biopharma announced the pricing of an underwritten public offering of 7,868,383 shares of common stock at $13.98 per share, with expected gross proceeds of approximately $110.0M before expenses. The clinical-stage company develops site-specific and novel-format ADCs. Participating investors included both new and existing healthcare-focused funds. Sutro intends to use the net proceeds, together with existing cash and equivalents, primarily for general corporate purposes, including research, clinical, and process development, manufacturing of product candidates, working capital, potential acquisitions or investments, and capital expenditures.
ADC Therapeutics reported preliminary Q4 and full year 2025 revenue and outlined upcoming clinical milestones [32]
ADC Therapeutics reported preliminary unaudited 2025 ZYNLONTA® net product revenue of approximately $73M, including $22M in Q4, and cash and cash equivalents of approximately $261M as of December 31, 2025, supporting an expected cash runway to at least 2028. Updated Phase 1b LOTIS-7 data showed an 89.8% overall response rate (ORR) and 77.6% complete response rate in 49 evaluable patients with relapsed or refractory diffuse large B cell lymphoma treated with ZYNLONTA plus glofitamab. Complete enrollment in LOTIS-7 is expected in 1H 2026. Topline results from the Phase 3 LOTIS-5 trial of ZYNLONTA plus rituximab in second-line or later DLBCL are anticipated in Q2 2026. IND-enabling activities were also completed for a prostate-specific membrane antigen-targeting ADC.
Merck completed $221.50-per-share acquisition of Cidara Therapeutics [33]
Merck announced the completion of its cash tender offer to acquire Cidara Therapeutics at $221.50 per share, with approximately 85.96% of outstanding shares tendered. Cidara will become a wholly owned subsidiary of Merck, and the transaction is expected to be accounted for as an asset acquisition, increasing 2026 research and development expenses by approximately $9B. The acquisition centers on CD388, an investigational drug–Fc conjugate comprising multiple copies of a small molecule neuraminidase inhibitor conjugated to a proprietary human antibody Fc fragment. Designed as a long-acting antiviral for influenza prevention, CD388 has shown broad preclinical activity against influenza A and B strains and is currently being evaluated in the Phase 3 ANCHOR study in high-risk adult and adolescent populations.
 |
Merck completed $221.50-per-share acquisition of Cidara Therapeutics; Market Trends. Credit: depositphotos.com |
RESEARCH AND DEVELOPMENT HIGHLIGHTS |
Wistar researchers developed HSP90-targeted small molecule drug conjugate to enhance AURKA inhibitor delivery [34]
Wistar Institute researchers reported development of a chimeric small molecule-drug conjugate linking an Aurora kinase A (AURKA) inhibitor to an HSP90-binding moiety to enhance tumor-selective delivery. The AURKA inhibitor component has shown clinical promise but is limited by toxicity. By conjugating it to an HSP90-targeting molecule, the researchers aimed to increase tumor accumulation, as HSP90 is overexpressed in cancer cells. In preclinical models, the conjugate bound both targets, inhibited cancer cell proliferation across multiple tumor types, and achieved up to tenfold higher tumor concentrations compared to the unconjugated inhibitor. The compound demonstrated prolonged tumor retention and favorable tolerability. Combination studies with a WEE1 inhibitor further enhanced tumor growth control.
Akari filed US provisional patent for CEACAM5-targeted ADC AKTX-102 with RNA-splicing payload [35]
Akari Therapeutics announced the filing of a US provisional patent application (No. 63/958,508) covering AKTX-102, a CEACAM5-targeted ADC. AKTX-102 combines a novel CEACAM5-directed antibody construct with the company’s proprietary PH1 spliceosome-modulating payload, designed to provide cytotoxic activity alongside innate and adaptive immune activation. The patent includes composition-of-matter claims for the antibody design and ADC constructs. CEACAM5 is broadly expressed across gastrointestinal, lung, bladder, and luminal A breast cancers, but has historically been challenging to target due to antigen shedding and soluble forms. The filing expands Akari’s intellectual property estate around its PH1 payload platform and ADC architecture, complementing its lead Trop2-directed program, AKTX-101, which is progressing toward investigational new drug submission.
Tubulis publishes Alco5 phosphoramidate linker platform enabling delivery of hydroxy-containing ADC payloads [36]
Tubulis has published preclinical proof-of-concept data in Nature Communications describing its Alco5 phosphoramidate linker technology, designed to expand the payload spectrum of ADCs to include hydroxy-containing drugs. The self-immolative phosphoramidate chemistry enables stable conjugation of structurally diverse alcohol-containing payloads, which are tracelessly released in the cytosol. In preclinical studies, Alco5 was applied to ten hydroxy-containing antiproliferative agents, including nucleoside analogues and elongation factor inhibitors, demonstrating sub-nanomolar potency retention, strong in vivo anti-tumor activity, high and homogeneous DAR, and favorable pharmacokinetics. By enabling access to novel MOAs such as protein degradation and overcoming the traditional payload limitations of tubulin and TOP1 inhibitors, Alco5 represents a significant advance in ADC linker technology and payload diversification.
Rakuten Medical expands academic access to IR700 dye under flexible research and IP terms [37]
Rakuten Medical announced it will provide IRDye® 700DX NHS ester (IR700 dye), a key component of its Alluminox® photoimmunotherapy platform, to academic researchers at an administrative fee-only acquisition cost. The initiative includes increased flexibility for publication, intellectual property rights, and potential commercialization opportunities under a Material Transfer Agreement. IR700 dye, a hydrophilic, light-activatable phthalocyanine derivative, is conjugated to targeting moieties in bioconjugate therapeutics, including Akalux™, approved in Japan for head and neck cancer. Rakuten Medical retains IP protection covering manufacturing and dye-conjugate compositions and will continue global supply. The program aims to accelerate innovation in light-activated bioconjugates and expand translational research applications beyond oncology.
 |
Rakuten Medical expands academic access to IR700 dye under flexible research and IP terms; Research and Development Highlights. Credit: www.istockphoto.com |
COMPANY START-UPS |
Fortitude Biomedicines launched with $13M seed financing to advance molecular glue-enabled ADC platform [38]
Fortitude Biomedicines launched following the close of a $13M seed financing co-led by K2 Bio Partners, Shanghai Healthcare Angel Capital, and Elikon Venture. Proceeds will support its lead immune cell-targeting biologic through IND-enabling studies and advance additional programs derived from its GLUE-DACTM platform. GLUE-DACTM is an ADC platform that incorporates proprietary molecular glue payloads, designed to combine antibody targeting with targeted protein degradation mechanisms. The company aims to develop ADCs for oncology and autoimmune diseases, including applications addressing resistance to conventional payloads. Fortitude was co-founded by Jesse Chen and Jin Wang, with the platform originating from research at Baylor College of Medicine.
CLINICAL TRIALS AND RESEARCH |
Innovent dosed first patient in Phase 3 trial of HER2 ADC in first-line metastatic breast cancer [39]
Innovent Biologics announced first patient dosing in the pivotal Phase 3 HeriCare-Breast01 trial evaluating IBI354, a HER2 monoclonal antibody–camptothecin derivative conjugate, as first-line treatment for unresectable locally advanced or metastatic HER2-positive breast cancer. The randomized, open-label study compares IBI354, with or without pertuzumab, versus paclitaxel plus trastuzumab and pertuzumab, with progression-free survival as the primary endpoint. In a prior Phase 1/2 study, IBI354 achieved a confirmed objective response rate of 59.1% overall and 72.4% at 9mg/kg every 3 weeks, with median progression-free survival of 14.1 months in that cohort. Grade ≥3 treatment-related adverse events occurred in 21.0% at 9mg/kg, with low incidence of interstitial lung disease (1.2%).
Phase 1/2 CADENZA data showed high response rates with CD123 ADC pivekimab sunirine in blastic plasmacytoid dendritic cell neoplasm [40]
Results from the Phase I/II CADENZA trial evaluating pivekimab sunirine (PVEK), a CD123-targeted ADC, in blastic plasmacytoid dendritic cell neoplasm were published in the Journal of Clinical Oncology. Among 33 frontline-treated patients, the overall response rate was 85%, with 75% achieving complete response and median overall survival of 16.6 months; eight patients proceeded to stem cell transplant. In 51 patients with relapsed or refractory disease, the overall response rate was 35% and median overall survival was 5.8 months. Treatment-related adverse events, including peripheral edema and infusion reactions, were reported as manageable. The data support further development of the CD123-directed ADC in this rare hematologic malignancy.
Henlius dosed first patient in Phase 1b/2 trial of PD-L1 ADC combinations in metastatic colorectal cancer [41]
Shanghai Henlius Biotech announced first patient dosing in China in the Phase 1b/2 trial (HLX43-mCRC202) evaluating its programmed PD-L1-targeting ADC HLX43 in combination with either the anti-EGFR monoclonal antibody HLX07 or the anti-PD-1 monoclonal antibody serplulimab in advanced or metastatic colorectal cancer. The study includes dose-escalation and expansion cohorts assessing safety, pharmacokinetics, immunogenicity, and efficacy. HLX43 incorporates a cleavable linker-payload licensed from MedLink’s TMALIN® platform and is designed to combine immune checkpoint blockade with payload-mediated cytotoxicity. The trial forms part of a broader global clinical program exploring HLX43 across multiple solid tumors, including NSCLC, cervical, and esophageal cancers.
Vaxcyte dosed first participants in Phase 3 OPUS-3 trial of 31-valent pneumococcal conjugate vaccine [42]
Vaxcyte announced first participant dosing in the Phase 3 OPUS-3 trial evaluating VAX-31, a 31-valent pneumococcal conjugate vaccine, in adults aged 50 years and older previously vaccinated against pneumococcal disease. The randomized, double-blind study (n~720) will assess safety, tolerability, and immunogenicity, including opsonophagocytic activity geometric mean titers across 31 serotypes plus 20B. OPUS-3 forms part of a broader Phase 3 program (OPUS-1, OPUS-2, OPUS-3) enrolling approximately 6,000 adults to support a planned BLA. VAX-31 is designed to expand serotype coverage relative to current standard-of-care vaccines. Topline data from OPUS-1 are expected in the Q4 of 2026, with OPUS-2 and OPUS-3 results anticipated in the first half of 2027.
 |
Vaxcyte dosed first participants in Phase 3 OPUS-3 trial of 31-valent pneumococcal conjugate vaccine; Clinical Trials and Research. Credit: www.istockphoto.com |
First patient dosed in Phase 1/2 trial of CD37-directed DXd ADC in B cell lymphoma [43]
Daiichi Sankyo announced first patient dosing in a first-in-human Phase 1/2 trial of DS3790, a CD37-directed DXd ADC, in relapsed or refractory B cell non-Hodgkin lymphoma. DS3790 comprises a humanized anti-CD37 IgG1 monoclonal antibody linked via tetrapeptide-based cleavable linkers to a TOP1 inhibitor payload (exatecan derivative, DXd). The multicenter, open-label study will enroll approximately 420 patients globally. The dose-escalation phase will evaluate DS3790 monotherapy to determine the recommended expansion dose, followed by dose-expansion cohorts and planned combination cohorts with other targeted therapies. Endpoints include safety, pharmacokinetics, biomarkers, and efficacy measures such as overall response rate, progression-free survival, and overall survival.
First patient dosed in Japan registrational Phase 2 trial of 177Lu-PSMA-I&T radioconjugate [44]
Curium Group, PeptiDream, and PDRadiopharma announced first patient dosing in a registrational Phase 2 trial in Japan evaluating 177Lu-PSMA-I&T for prostate-specific membrane antigen (PSMA)-positive metastatic castration-resistant prostate cancer. 177Lu-PSMA-I&T is a PSMA-targeting ligand conjugated to a chelator radiolabeled with Lutetium-177. The multicenter, open-label, single-arm study will assess efficacy and safety, using bridging data from Curium’s global clinical program. A companion diagnostic trial of 64Cu-PSMA-I&T, a PET imaging agent, is also underway in Japan and is used to confirm PSMA positivity prior to therapy. Under the collaboration, PDRadiopharma will lead regulatory, manufacturing, and commercialization activities in Japan, with Curium supporting technology transfer and global development.
Avacta updated Phase 1 protocol for faridoxorubicin peptide drug conjugate AVA6000 [45]
Avacta Therapeutics announced protocol updates to the Phase 1 clinical program for faridoxorubicin (AVA6000), a FAPα-cleavable peptide–doxorubicin conjugate developed using the pre|CISION platform. The maximum cumulative dosing limit based on exposure to released doxorubicin has been removed following favorable safety data, including escalation to approximately four times the conventional doxorubicin dose and cumulative exposure of 550 mg/m² without severe cardiac toxicity. The protocol also allows greater flexibility in dose levels, with final Phase 1b cohorts comparing two dose levels to determine the optimal biologic dose for future efficacy studies.
Vaxcyte advanced Phase 3 and Phase 2 programs for 31-valent pneumococcal conjugate vaccine VAX-31 [46]
Vaxcyte announced clinical progress for VAX-31, its 31-valent pneumococcal conjugate vaccine (PCV) candidate. In adults aged 50 years and older, the first participants were dosed in the Phase 3 OPUS-2 trial evaluating concomitant administration with a licensed seasonal influenza vaccine. A second Phase 3 study (OPUS-3) in previously pneumococcal-vaccinated adults is planned for the first quarter of 2026, while the pivotal noninferiority OPUS-1 trial remains ongoing. In the pediatric program, enrollment has been completed in a Phase 2 dose-finding study in more than 900 healthy infants. Topline safety, tolerability, and immunogenicity data from the adult and infant studies are expected between Q4 of 2026 and the first half of 2027, supporting a planned BLA submission.
Mabwell dosed first US patient in Phase 1/2 trial of CDH17-targeting ADC [47]
Mabwell announced first patient dosing in a US Phase 1/2 first-in-human trial (NCT07216560) of 7MW4911, a CDH17-targeting ADC, for advanced colorectal cancer and other gastrointestinal tumors. The study will evaluate safety, pharmacokinetics, and efficacy. 7MW4911 comprises a CDH17-specific monoclonal antibody, a cleavable linker with high plasma stability, and a proprietary DNA TOP1 inhibitor payload (MF-6) designed to address multidrug resistance. The candidate was developed using Mabwell’s IDDC™ platform and has previously entered clinical evaluation in China. Preclinical data in CDX and PDX gastrointestinal tumor models showed antitumor activity, including activity in multidrug-resistant models compared with MMAE- and DXd-based ADCs.
First patient dosed in Phase 3 TROPION-Lung17 trial of TROP2-directed ADC DATROWAY in NSCLC [48]
The first patient was dosed in the Phase 3 TROPION-Lung17 trial evaluating DATROWAY® (datopotamab deruxtecan) versus docetaxel in patients with TROP2 normalized membrane ratio (NMR)-positive, locally advanced or metastatic nonsquamous NSCLC without actionable genomic alterations following prior immunotherapy and platinum-based chemotherapy. DATROWAY is a TROP2-directed ADC comprising a humanized anti-TROP2 IgG1 linked via a cleavable tetrapeptide linker to a TOP1 inhibitor payload (DXd). The global study will enroll ~400 patients and prospectively select participants using an investigational AI-enabled companion diagnostic. Dual primary endpoints are progression-free survival and overall survival. DATROWAY is jointly developed by Daiichi Sankyo and AstraZeneca.
 |
First patient dosed in Phase 3 TROPION-Lung17 trial of TROP2-directed ADC DATROWAY in NSCLC48; Clinical Trials and Research. Credit: datroway.com |
Kivu Bioscience dosed first patient in Phase 1 trial of PTK7-targeted ADC KIVU-107 [49]
Kivu Bioscience announced dosing of the first patient in a Phase 1 first-in-human trial of KIVU-107, a PTK7-targeted ADC, in advanced solid tumors. The study (NCT07229313) will evaluate safety, tolerability, pharmacokinetics, and preliminary antitumor activity. KIVU-107 incorporates GlycoConnect® and HydraSpace® site-specific conjugation technologies to enable defined DAR and optimized pharmacokinetics, to expand the therapeutic window. The program advanced from preclinical development to clinical dosing in under 12 months. The trial has received regulatory approval in the US and Australia and is enrolling patients with PTK7-expressing advanced solid malignancies.
Henlius presented Phase 2 proof-of-concept data for PD-L1 ADC in pretreated ESCC at ASCO GI 2026 [50]
Henlius presented Phase 2 proof-of-concept data for HLX43, a PD-L1-targeted ADC, in recurrent or metastatic esophageal squamous cell carcinoma (ESCC) at ASCO GI 2026. In the randomized study (n=37), patients received 2, 2.5, or 3 mg/kg every 3 weeks after prior chemoimmunotherapy. Among 33 response-evaluable patients, ORR was 30.3% and disease control rate (DCR) was 81.8%. In the 3 mg/kg cohort (n=13), ORR reached 61.5%, DCR 100%, and confirmed ORR 38.5%. Grade ≥3 treatment-related adverse events occurred in 29.7% overall. HLX43 integrates immune checkpoint blockade with cytotoxic payload delivery and is being evaluated across multiple solid tumors.
ALX Oncology advanced Phase 1 EGFR-targeted ADC and dosed first patient in Phase 2 trial [51]
ALX Oncology announced dosing of the first patient in the Phase 2 ASPEN-09-Breast trial evaluating the CD47 inhibitor evorpacept in combination with trastuzumab and chemotherapy in HER2-positive metastatic breast cancer previously treated with fam-T-DXd. The company also reported progress in its Phase 1 trial of ALX2004, an EGFR-targeted ADC, which has advanced to the third dose cohort (4 mg/kg) following no dose-limiting toxicities in earlier cohorts. ALX2004 is being evaluated in advanced or metastatic EGFR-expressing solid tumors. The Phase 1 study includes dose escalation and expansion, with an initial safety update expected in 1H 2026.
Virometix reported positive Phase 1 data for synthetic peptide–SVLP conjugate vaccine V-212 [52]
Virometix announced positive topline results from a randomized, double-blind, placebo-controlled Phase 1 trial of V-212, a fully synthetic, serotype-independent pneumococcal vaccine candidate. The study enrolled 60 healthy volunteers aged 18–45 and evaluated safety and immunogenicity across three dose levels. V-212 comprises conserved pneumococcal peptide epitopes conjugated to proprietary Synthetic Virus-Like Particle (SVLP) nanoparticles incorporating built-in adjuvant elements. The vaccine was well tolerated, with predominantly mild to moderate adverse events and no serious adverse events reported. Geometric mean IgG titers increased two- to six-fold over baseline across the three target epitopes, with evidence of a boosting effect after subsequent vaccinations. Results support further development of V-212 alone and in combination with approved pneumococcal conjugate vaccines.
Rani Therapeutics initiated Phase 1 trial of orally delivered Fc-fusion GLP-1/GLP-2 conjugate for the treatment of obesity [53]
Rani Therapeutics announced the initiation of a Phase 1 clinical trial evaluating RT-114, an orally administered RaniPill® capsule containing PG-102, an Fc-fusion protein conjugated GLP-1/GLP-2 dual agonist developed by ProGen. The study will assess safety, tolerability, bioavailability, and PK/PD following single and multiple doses in obesity. PG-102 is designed as a dual agonist targeting GLP-1 and GLP-2 pathways, and prior preclinical and clinical data presented at EASD 2025 showed glucose-lowering effects and early weight-loss signals. In animal models, oral delivery demonstrated bioavailability, pharmacokinetics, and weight loss comparable to subcutaneous administration. The trial marks clinical evaluation of the Fc-fusion conjugate delivered through an oral biologics platform.
TOOLS AND TECHNOLOGIES |
Decoy Therapeutics advanced manufacturing platform for peptide-conjugate antivirals under Gates Foundation agreement [54]
Decoy Therapeutics announced development of a globally accessible manufacturing platform for peptide-conjugate antivirals as part of its funded Global Access Commitment Agreement with the Gates Foundation. The platform is designed to enable rapid, cost-efficient scale-up of peptide-conjugate fusion inhibitors using standard peptide synthesis equipment across a distributed manufacturing network. Built on Decoy’s IMP3ACT computational design and manufacturing platform, the approach integrates machine learning-enabled peptide engineering with lipid conjugation to enhance pharmacokinetics and scalability. The capability will be validated using an intranasal pan-coronavirus fusion inhibitor and is intended to support future peptide drug conjugate programs, including oncology indications, while facilitating global access in low- and middle-income countries.
 |
| Decoy Therapeutics advanced manufacturing platform for peptide-conjugate antivirals under Gates Foundation agreement; Tools and Technologies. Credit: www.istockphoto.com |
CONFERENCES, EVENTS, AND PUBLICATIONS |
Precision Biologics present preclinical data on glycan-targeting ADC at Ace Drug Discovery Summit [55]
Precision Biologics announced that its CEO presented preclinical data on PB-223 (PB-vcMMAE-5), a tumor-specific ADC, at the 6th Ace Drug Discovery Summit in London. PB-223 comprises a monoclonal antibody targeting truncated core 2 O-glycans conjugated to a monomethyl auristatin E payload via a valine–citrulline linker. The presentation described in vitro and in vivo efficacy data demonstrating tumor-selective activity across multiple human solid tumor models, including complete tumor eradication in xenograft models with minimal observed toxicity in preclinical studies. The target antigen is reported to be expressed broadly in solid tumors but not in healthy tissue.
OKYO to present Phase 2 data on lipid-conjugated peptide urcosimod at ASCRS 2026 [56]
OKYO Pharma announced that an abstract on urcosimod, a lipid-conjugated chemerin peptide agonist of the ChemR23 receptor, has been accepted for presentation at the 2026 American Society of Cataract and Refractive Surgery Annual Meeting. The presentation will report efficacy and safety data from a proof-of-concept Phase 2 study in neuropathic corneal pain. Urcosimod is designed to modulate inflammatory and neuronal pathways and has previously demonstrated statistically significant pain reduction in an 18-patient randomized, placebo-controlled Phase 2 trial. The candidate has also shown positive results in a 240-patient Phase 2 trial in dry eye disease and has received investigational new drug clearance and fast track designation from the FDA for neuropathic corneal pain.
Caris published multiomic analysis of resistance mechanisms to trastuzumab deruxtecan in metastatic breast cancer [57]
Caris Life Sciences published a study analyzing mechanisms of resistance to ENHERTU (T-DXd) in metastatic breast cancer using multiomic molecular profiling and real-world clinico-genomic data. The analysis included 2,799 T-DXd-treated patients and integrated whole exome sequencing and whole transcriptome sequencing. ERBB2 and ABCC1 expression were identified as key transcriptomic predictors of T-DXd–specific overall survival, with higher ERBB2 expression associated with improved outcomes and higher ABCC1 expression associated with poorer outcomes, independent of HER2 classification. Post-treatment samples showed increased ABCC1 expression and enrichment of mutations in ERBB2, NFE2L2, KEAP1, and TOP1. Preclinical models supported a role for ABCC1-mediated drug efflux in acquired resistance to the HER2-directed ADC.
Gilead Sciences published Phase 3 ASCENT-04 results for sacituzumab govitecan plus pembrolizumab [58]
Gilead Sciences announcedpublication of Phase 3 ASCENT-04/KEYNOTE-D19 results evaluating the Trop-2-directed ADC sacituzumab govitecan (Trodelvy) plus pembrolizumab in first-line PD-L1+ metastatic TNBC. The study met its primary endpoint, demonstrating a 35% reduction in risk of disease progression or death versus pembrolizumab plus chemotherapy (hazard ratio 0.65; p<0.001). Median progression-free survival was 11.2 months with the combination (n=221) versus 7.8 months in the control arm (n=222). Grade ≥3 adverse events included neutropenia (43%) and diarrhea (10%) in the combination arm. Supplemental applications have been submitted to the FDA and EMA for the investigational first-line indications.
Aro Biotherapeutics to present Phase 1b data for CD71-targeted siRNA conjugate ABX1100 at WORLD Symposium 2026 [59]
Aro Biotherapeutics announced it will present interim Phase 1b clinical data for ABX1100 at the 22nd Annual WORLD Symposium. ABX1100 is a CD71-targeting Centyrin–siRNA conjugate designed to silence GYS1 expression in muscle tissue for the treatment of late-onset Pompe disease. The oral presentation will include preliminary safety, tolerability, biomarker, and pharmacodynamic data in patients receiving enzyme replacement therapy. ABX1100 represents the first clinical evidence of a GYS1-targeted substrate reduction strategy using a tissue-targeted siRNA conjugate. The investigational therapy has received Orphan Drug Designation and Rare Pediatric Disease status from the FDA .
Antengene presented Phase 1/2 data for CLDN18.2-targeted ADC ATG-022 and pipeline updates at JPM 2026 [60]
Antengene presented updated Phase 1/2 CLINCH data for ATG-022, a CLDN18.2-targeted ADC, at the 44th Annual J.P. Morgan Healthcare Conference. In moderate-to-high CLDN18.2–expressing gastric cancer, objective response rates reached 40% (2.4 mg/kg) and 46.7% (1.8 mg/kg), with manageable safety (Grade ≥3 TRAEs 19.4% at 1.8 mg/kg). Activity was also observed in low-expression and non-gastrointestinal tumors. Antengene is advancing ATG-022 across first- to third-line gastric cancer settings. The company also highlighted ATG-125, a B7-H3 × PD-L1 bispecific ADC integrating cytotoxic and immuno-oncology mechanisms with an IND planned for Q1 2027, and progress across its AnTenGager™ T cell engager platform.
Lahjavida to present Dye-Drug Conjugate platform data at AACR 2026 [61]
Lahjavida Bio announced that an abstract describing its proprietary Dye-Drug Conjugate (DDC) platform has been accepted for presentation at the AACR Annual Meeting 2026. The company’s DDCs are small-molecule ‘dye–linker–payload’ constructs designed to improve therapeutic index through tumor-selective delivery, conceptually analogous to ADCs but leveraging dye chemistry instead of antibodies for targeting. The platform aims to enable selective tumor uptake of potent cytotoxic payloads using small-molecule conjugation strategies. Additional details remain under embargo in accordance with AACR policies. The presentation will highlight Lahjavida’s approach to tumor-targeted small-molecule conjugate therapeutics in oncology.
IDEAYA outlined 2026 objectives, including registrational plans for DLL3-targeted TOP1 ADC at J.P. Morgan conference [62]
IDEAYA Biosciences provided a business update at the 44th Annual J.P. Morgan Healthcare Conference outlining 2026 objectives, including advancement of its ADC pipeline. The company plans to initiate a registrational monotherapy study of IDE849, a DLL3-targeted TOP1 ADC, in second-line or refractory SCLC and/or neuroendocrine carcinomas by the end of 2026. IDEAYA also intends to begin a Phase 1 dose-escalation trial of IDE034, a B7H3/PTK7 bispecific TOP1 ADC, in Q1 2026, and to evaluate combination strategies with its DNA damage response assets. The company reported approximately $1.1B in cash as of September 30, 2025, supporting operations into 2030.
References
- Araris Biotech AG. Chugai and Araris Biotech AG enter license agreement for Araris’ linker-payload ADC technology AraLinQ. Feb 10, 2026.
- SK bioscience. SK bioscience and Sanofi Launch MenQuadfi Meningococcal Vaccine in Korea. Jan 9, 2026.
- Wave Life Science. Wave Life Sciences Announces Plans to Accelerate Regulatory Engagement with Full Control of WVE-006 for Alpha-1 Antitrypsin Deficiency. Feb 2, 2026.
- StarkAge Therapeutics. StarkAge Therapeutics Announces Research Collaboration with Gustave Roussy to Advance Senolytic Therapies in Digestive Cancers. Jan 27, 2026.
- HealZen Therapeutics. HealZen Announces Global Strategic Collaboration to Develop Degrader-Antibody Conjugate Therapeutics. Jan 26, 2026.
- Rakuten Medical. Rakuten Medical and LOTTE Biologics Sign Manufacturing Agreement to Support Biopharmaceuticals in Global Oncology Program. Jan 13, 2026.
- Tahoe Therapeutics. Tahoe Therapeutics and Alloy Therapeutics to Form Joint Venture and Develop First-in-Class Antibody-Drug Conjugates for Hard-to-Treat Cancers. Jan 13, 2026.
- Summit Therapeutics. Summit Therapeutics Announces Clinical Trial Collaboration with GSK to Evaluate Ivonescimab in Combination with GSK’s B7-H3 Antibody Drug Conjugate (ADC). Jan 12, 2026.
- AbbVie. AbbVie and RemeGen Announce Exclusive Licensing Agreement to Develop A Novel Bispecific Antibody for Advanced Solid Tumors. Jan 12, 2026.
- Biocytogen. Biocytogen and Acepodia Expand Collaboration Through Option-based Evaluation Framework for First-in-Class Bispecific and Dual-Payload ADCs (BsAD2C). Jan 9, 2026.
- MediLink Therapeutics. MediLink Has Signed an Additional Exclusive Licensing Agreement with Roche. Jan 9, 2026.
- InduPro Therapeutics. InduPro Therapeutics Announces Strategic Collaboration with Lilly to Develop First-in-Class Bispecific/Multispecific Oncology Therapeutics Using InduPro’s Proximity Guided Platform. Jan 7, 2026.
- DISCO Pharmaceutical. DISCO Pharmaceuticals Enters License Agreement to Advance Novel Cancer Surfaceome Targeted Therapies. Jan 7, 2026.
- Synaffix. Lonza’s Synaffix and Sidewinder Therapeutics Announce Multi-Target License Agreement to Advance Development of Next-Generation Bispecific ADCs. Jan 7, 2026.
- OBI Pharma. OBI Pharma and TegMine Therapeutics Sign Exclusive Global License Agreement for Glycan-Targeting ADC. Dec 19, 2025.
- Daiichi Sankyo. DATROWAY® Supplemental New Drug Application Submitted in Japan for Patients with Metastatic Triple Negative Breast Cancer Who Are Not Candidates for Immunotherapy. Feb 12, 2026.
- Astellas. Astellas Submits Supplemental New Drug Application in Japan for PADCEV™ (enfortumab vedotin) with Keytruda ® (pembrolizumab) for Certain Patients with Muscle-Invasive Bladder Cancer. Jan 30, 2026.
- Shanghai Henlius Biotech. Henlius Receives an IND Approval for Its PD-L1-Targeting ADC HLX43 in Combination with Anti-PD-1 mAb Serplulimab and Novel Anti-EGFR mAb HLX07. Jan 27, 2026.
- Daiichi Sankyo. ENHERTU® Approved in China as the First and Only HER2 Directed ADC for the Second-Line Treatment of Patients with HER2 Positive Metastatic Gastric Cancer. Jan 22, 2026.
- NEOK Bio. NEOK Bio Receives FDA IND Approval for NEOK001, A First-In-Class B7-H3/ROR1 Bispecific ADC for Treatment of Solid Tumor Cancers. Jan 21, 2026.
- Avacta Therapeutics. Avacta Announces U.S. Food and Drug Administration Clearance of the Investigational New Drug (IND) Application for the Second pre|CISION® Medicine, FAP-Exatecan (AVA6103). Jan 21, 2026.
- Otsuka Pharmaceutical. DawnzeraTM (donidalorsen) granted European Union marketing authorisation for the prevention of hereditary angioedema. Jan 21, 2026.
- BPGbio. BPGbio Receives FDA Orphan Drug Designation for BPM31510 for the Treatment of Primary CoQ10 Deficiency. Jan 20, 2026.
- Eli Lilly. Lilly’s sofetabart mipitecan receives U.S. FDA’s Breakthrough Therapy designation for the treatment of certain patients with platinum-resistant ovarian cancer. Jan 20, 2026.
- Dyne Therapeutics. Dyne Therapeutics Receives Orphan Drug Designation in Japan for Zeleciment Basivarsen (DYNE-101) for Myotonic Dystrophy Type 1. Jan 20, 2026.
- Daiichi Sankyo. ENHERTU® Plus Pertuzumab Type II Variation Application Validated in the EU as First-Line Treatment of Patients with HER2 Positive Metastatic Breast Cancer. Jan 19, 2026.
- Whitehawk Therapeutics. Whitehawk Therapeutics Advances to Clinical Stage with IND Clearance for HWK-007 and Announces IND Submission for HWK-016. Jan 8, 2026.
- Arbele. Arbele Receives FDA Orphan Drug Designation for ARB1002, an Anti-CDH17 Antibody–Drug Conjugate for the Treatment of Pancreatic Cancer. Jan 8, 2026.
- Kyntra Bio. FibroGen Rebrands as Kyntra Bio to Reflect a New Era of Focus and Momentum. Jan 08, 2026.
- Intensity Therapeutics. Intensity Therapeutics Announces Reverse Stock Split. Feb 13, 2026.
- Sutro Biopharma. Sutro Biopharma Announces Pricing of $110.0 million Underwritten Offering. Feb 10, 2025.
- ADC Therapeutics. ADC Therapeutics Provides Preliminary Fourth Quarter and Full Year 2025 Revenue and Cash Estimates and Recent Corporate Updates. Jan 8, 2026.
- Merck. Merck to Complete Acquisition of Cidara Therapeutics. Jan 7, 2026.
- The Wistar Institute. Dual-Action Molecule Design Concentrates Cancer Treatment in Tumor Cells to Allow Higher Doses. gEB 6, 2026.
- Akari Therapeutics. Akari Therapeutics Files Key Patent and Unveils Second ADC Program AKTX-102 Targeting CEACAM5 Expressing Solid Tumors. Jan 26, 2026.
- Tubulis. Tubulis Unveils Alco5 Conjugation Platform’s Ability to Expand ADC Payload Scope in Nature Communications. Jan 21, 2026.
- Rakuten Medical. Rakuten Medical Expands Academic Access to IR700 Dye Through Fee-Only Provision, Greater Publication Freedom and Expanded IP & Commercialization Opportunities. Jan 20, 2026.
- Fortitude Biomedicines. Fortitude Biomedicines Launches With $13M in Financing to Advance Novel Antibody-based Therapies for Treatment of Autoimmune Diseases and Cancer. Jan 26, 2026.
- Innovent Biologics. Innovent Dosed First Participant in Phase 3 Clinical Study of IBI354 (Novel HER2 ADC) for First Line Treatment of HER2-positive Breast Cancer. Feb 13, 2026.
- The University of Texas, MD Anderson Cancer Center. Antibody-Drug Conjugate Achieves High Response Rates as Frontline Treatment in Aggressive, Rare Blood Cancer. Feb 11, 2026.
- Shanghai Henlius Biotech. First Patient Dosed for a Phase 1b/2 Clinical Trial of PD-L1 ADC HLX43 Combination Therapy in Advanced Colorectal Cancer. Feb 11, 2026.
- Vaxcyte. Vaxcyte Doses First Participants in OPUS-3 Phase 3 Trial Evaluating VAX-31 in Adults Previously Vaccinated with Lower-Valency Pneumococcal Vaccines. Feb 11, 2026.
- Daiichi Sankyo. DS3790 Enters Clinical Development as First DXd ADC in Hematology from Industry-Leading ADC Portfolio of Daiichi Sankyo. Feb 4, 2026.
- Curium Pharma. Curium Group, PeptiDream and PDRadiopharma Enroll First Patient to Registrational Clinical Trial of 177Lu-PSMA-I&T for Prostate Cancer in Japan. Feb 5, 2026.
- Avacta Therapeutics. Avacta announces two key clinical updates to its faridoxorubicin program. Feb 3, 2026.
- Vaxcyte. Vaxcyte Advances Adult and Infant Programs for VAX-31, a Potential Best-in-Class, Next-Generation Pneumococcal Conjugate Vaccine. Jan 22, 2026.
- Mabwell. Mabwell Announces First-Patient Dosed in Clinical Trial of Novel CDH17-Targeting ADC 7MW4911 in the U.S. Jan 21, 2026.
- Daiichi Sankyo. TROPION-Lung17 TROP2 Biomarker Directed Phase 3 Trial of DATROWAY® Initiated in Patients with Previously Treated Advanced Nonsquamous Non-Small Cell Lung Cancer. Jan 13, 2026.
- Kivu Bioscience. Kivu Bioscience Announces First Patient Dosed in Phase 1 Trial of KIVU-107 Next-Generation PTK7-Targeting ADC for Advanced Solid Tumors. Jan 12, 2026.
- Henlius. Henlius Debuts Phase 2 Results for Its Broad Anti-tumor PD-L1 ADC HLX43 in Second-Line or Subsequent Treatment of Esophageal Squamous Cell Carcinoma at ASCO GI 2026. Jan 09, 2026.
- ALX Oncology. ALX Oncology Advances Separate Clinical Trials Evaluating Investigational CD47-Inhibitor Evorpacept and Novel EGFR-Targeted Antibody-Drug Conjugate ALX2004. Jan 8, 2026.
- Virometix Vaccines. Virometix AG Announces Positive Data from Phase 1 Trial of V-212, a Fully Synthetic, Serotype-Independent Vaccine Development Candidate for the Prevention of Pneumococcal Disease. Jan 8, 2026.
- Rani Therapeutics. Rani Therapeutics Initiates Phase 1 Study of RT-114 RaniPill® for the Treatment of Obesity in Collaboration with ProGen. Jan 8, 2026.
- Decoy Therapeutics. Decoy Therapeutics Reaches Global Access Commitment Agreement to Focus on Development of a Globally Accessible, Scalable Peptide-Conjugate Manufacturing Platform. Jan 13, 2026.
- Precision Biologics. PB-223, A Novel Antibody Drug Conjugate Targeting Truncated Core-2 Glycans in Solid Tumors. Feb 19, 2026.
- OKYO Pharma. OKYO Pharma Announces Acceptance of Urcosimod Phase 2 Study Results for Presentation at Prestigious ASCRS Annual Meeting. Jan 30, 2026.
- Caris Lide Sciences. Caris Life Sciences’ Real-World Data Uncovers Metastatic Breast Cancer Patient Responses and Resistance to Trastuzumab Deruxtecan. Jan 28, 2026.
- Gilead Sciences. New England Journal of Medicine Publishes Phase 3 ASCENT-04/KEYNOTE-D19 Results Supporting Trodelvy® Plus Keytruda® as a Potential New Standard of Care in First-line PD-L1+ Metastatic Triple-negative Breast Cancer. Jan 21, 2026.
- Aro Biotherapeutics. Aro Biotherapeutics to Present Clinical Update from its Pompe Disease Program at the 22nd Annual WORLD Symposium. Jan 20, 2026.
- Antegene. Antengene Presents at JPM: Strong Clinical Data Update and Strategic Focus on Next-Generation ADCs and TCEs. Jan 16, 2026.
- Lahjavida. Lahjavida Bio Accepted to Present Tumor-Targeted Small-Molecule Drug Delivery Platform at AACR Annual Meeting 2026. Jan 14, 2026.
- IDEAYA Bioscience. IDEAYA Biosciences Provides a Business Update and Outlines 2026 Corporate Objectives at the 44th Annual J.P. Morgan Healthcare Conference. Jan 11, 2026.

